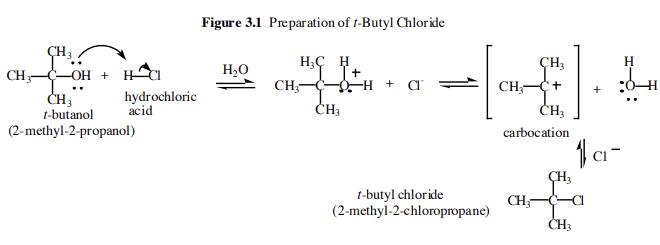
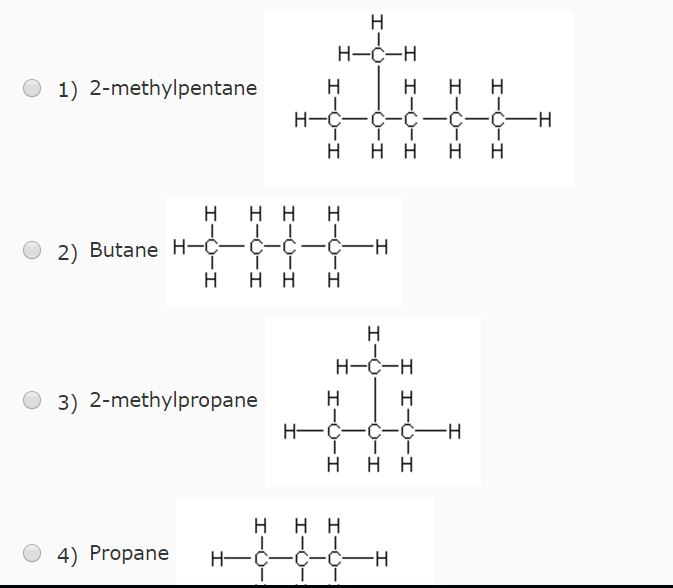
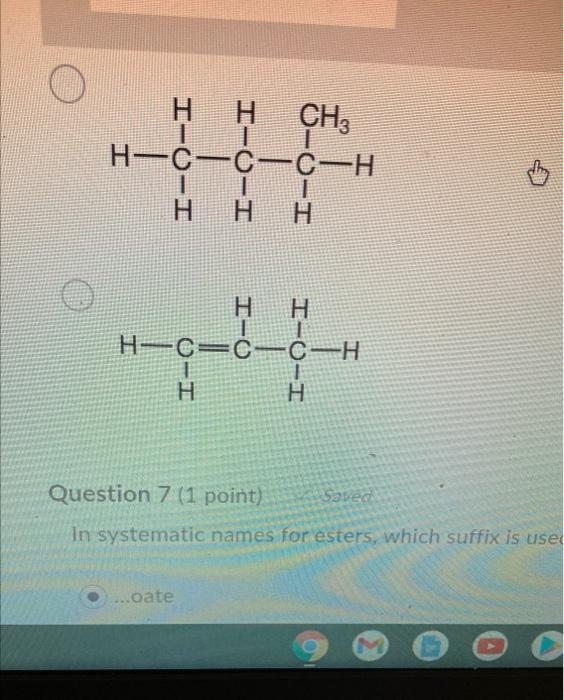
Arrange the following in decreasing order of their boiling points.(I) n - Butane (II) 2 - Methylbutane(III) n - Pentane(IV) 2,2 - Dimethylpropane

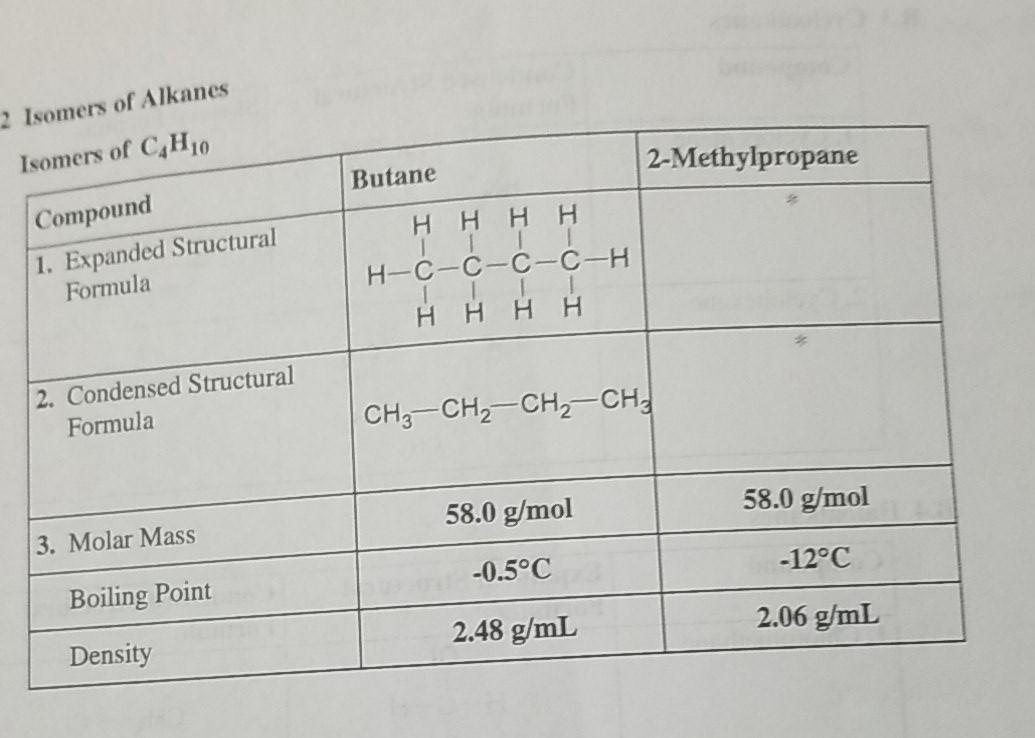
SOLVED: Question 23 (a) Butane and 2-methylpropane are both non-polar and have the same molecular formula,C4H1o. Explain why butane has a boiling point of -0.5-C and 2-methylpropane has a boiling point of -
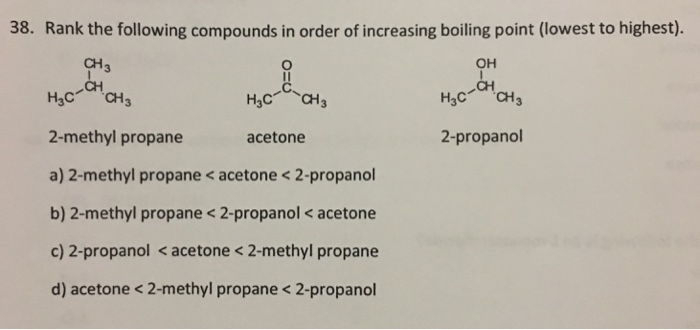
32.Which of the following has the lowest melting point? A)2 methyl butane B)2 methyl propane C)2,2 dimethyl propane D)n pentane
![SOLVED: Problem 11.23 Butane and 2-methylpropane whose space-filling models are shown are both nonpolar and have the same molecular formula yet butane has the higher boiling point] (0.5 C compared t0 -11.7 SOLVED: Problem 11.23 Butane and 2-methylpropane whose space-filling models are shown are both nonpolar and have the same molecular formula yet butane has the higher boiling point] (0.5 C compared t0 -11.7](https://cdn.numerade.com/ask_previews/558c9c9f-69dc-4898-b90e-97ee9014db79_large.jpg)
SOLVED: Problem 11.23 Butane and 2-methylpropane whose space-filling models are shown are both nonpolar and have the same molecular formula yet butane has the higher boiling point] (0.5 C compared t0 -11.7