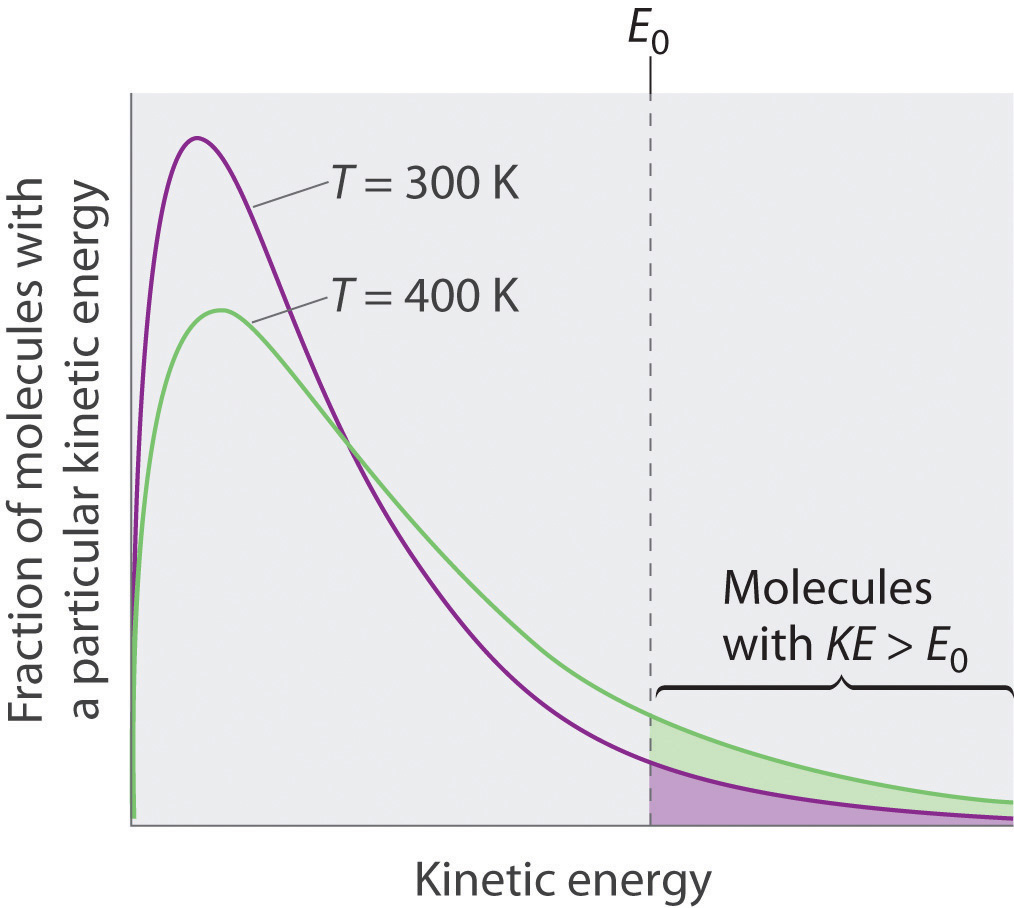
Is a 50/50 mixture of acetone and water an azeotrope? Also, why does the first drop of destillate form at 40C? - Quora
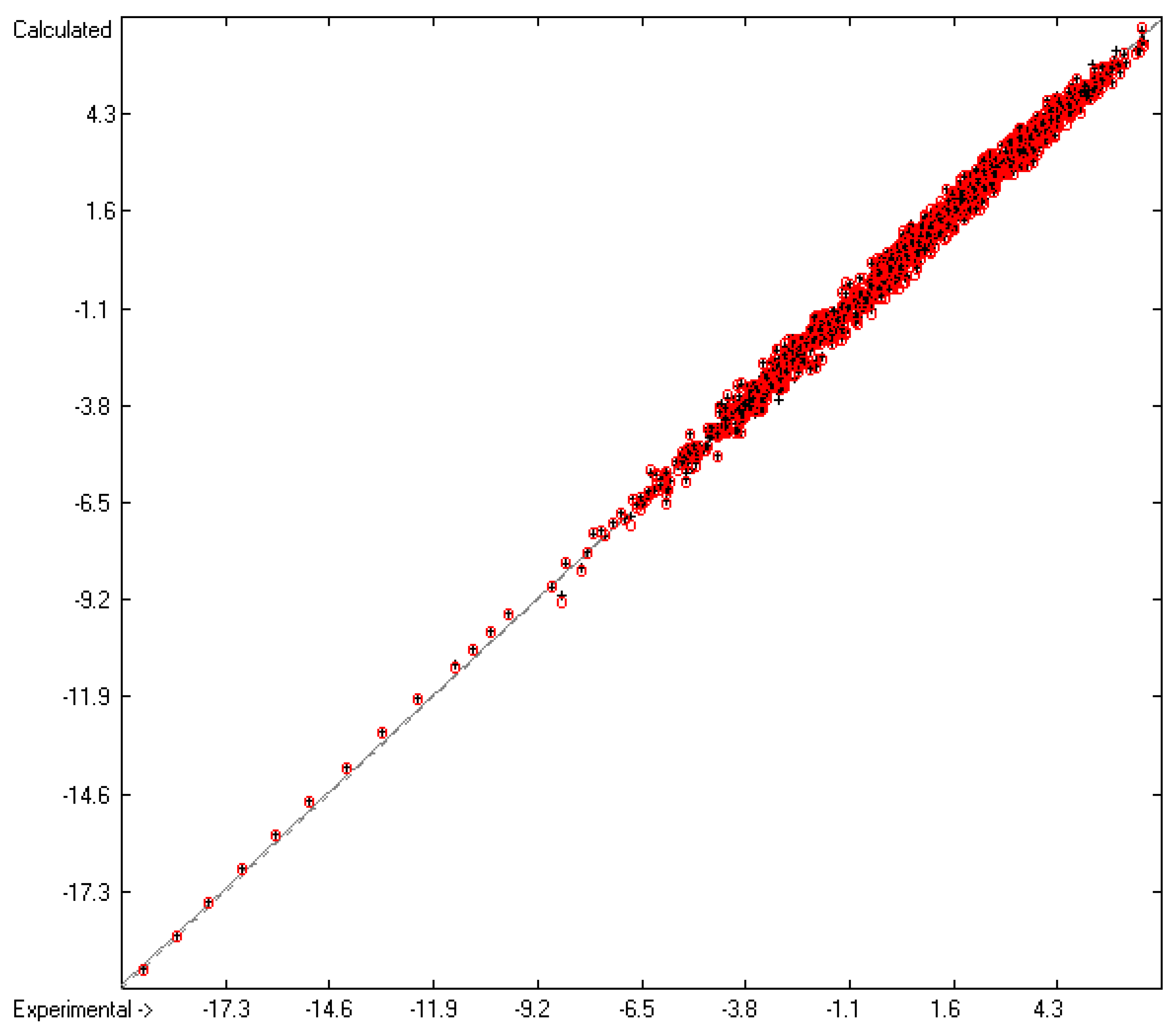
Molecules | Free Full-Text | Calculation of the Vapour Pressure of Organic Molecules by Means of a Group-Additivity Method and Their Resultant Gibbs Free Energy and Entropy of Vaporization at 298.15 K

If the normal boiling point of acetone is 56^∘ C and it has a Δ H^∘vap of 32.1 kJ/mol, estimate the boiling point at 5 bar?
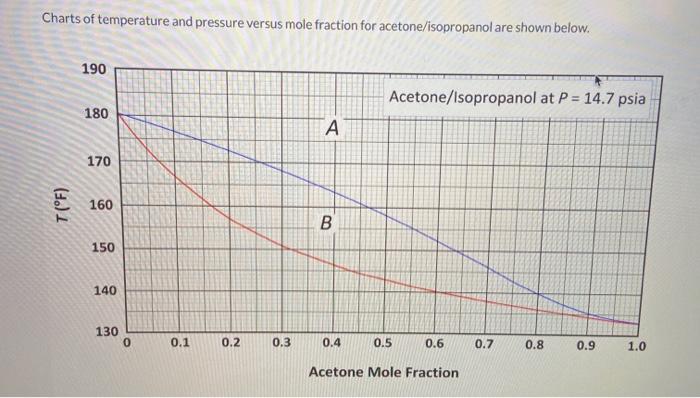
How to calculate the boiling point of a mixture made up of 2 liquids ( acetone+water in the same ratios) - Quora
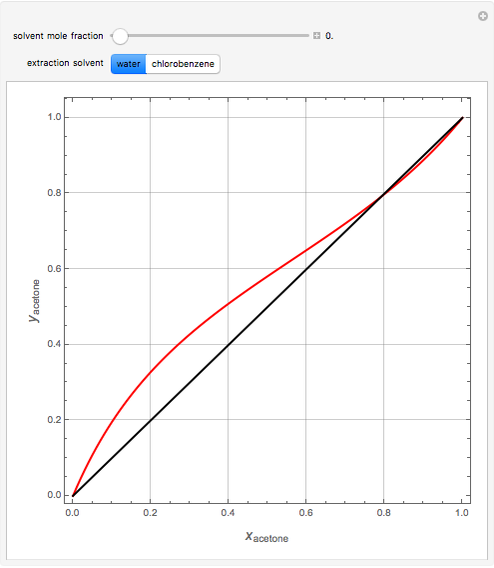
Breaking the Acetone-Methanol Azeotrope with Different Extraction Solvents - Wolfram Demonstrations Project

Use the following vapor pressure diagram to estimate the partial pressure of acetone. | Homework.Study.com
Equilibrium phase diagram of acetone/ethanol mixture under the pressure... | Download Scientific Diagram

Comparison of extractive distillation and pressure-swing distillation for acetone/chloroform separation - ScienceDirect