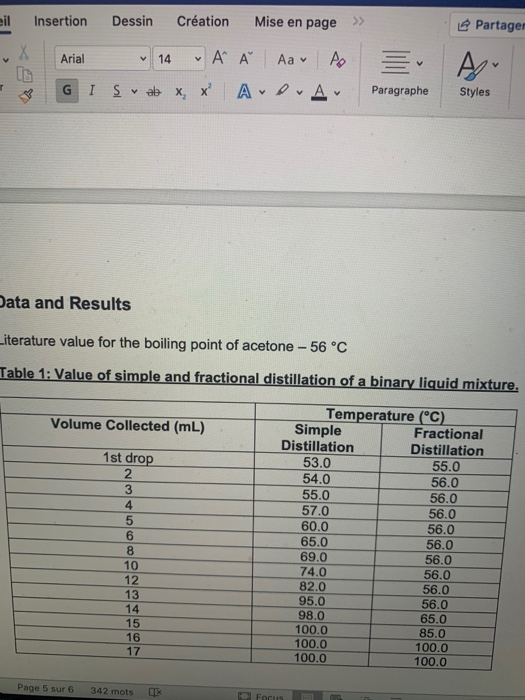
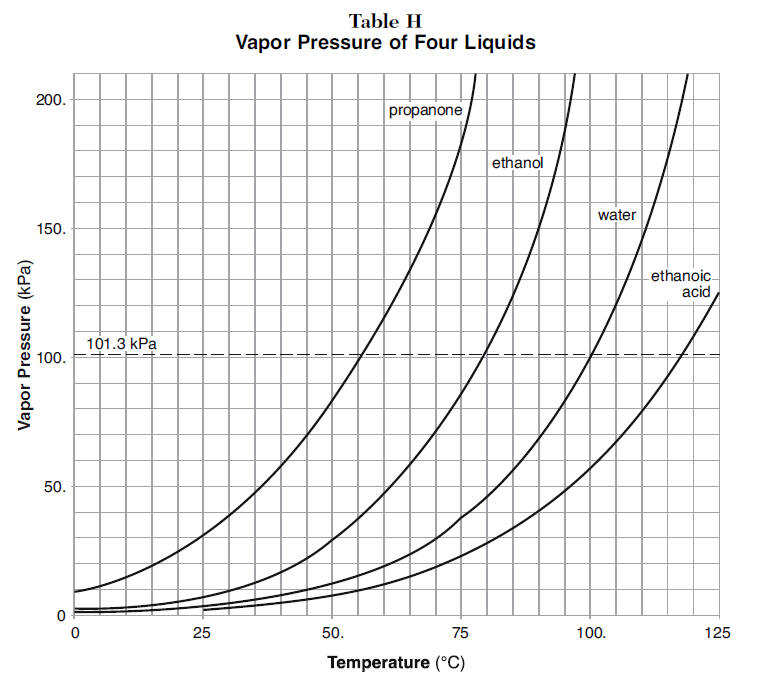
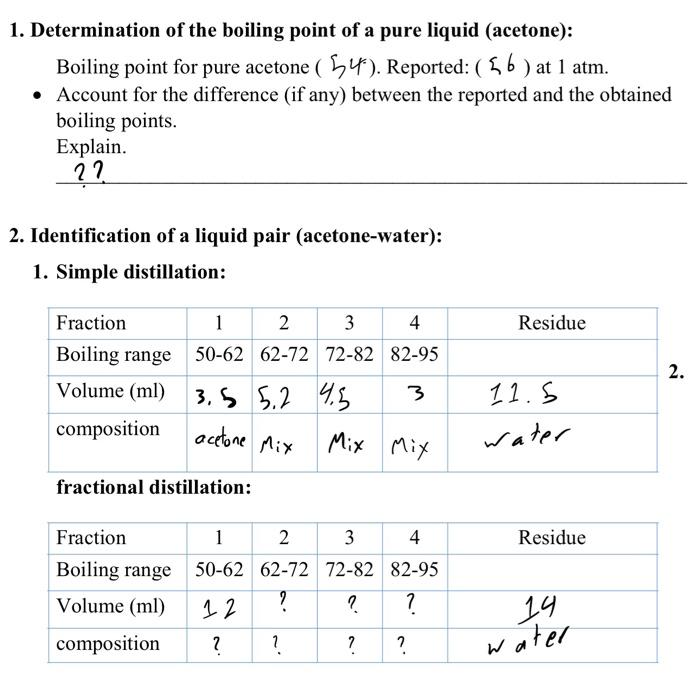
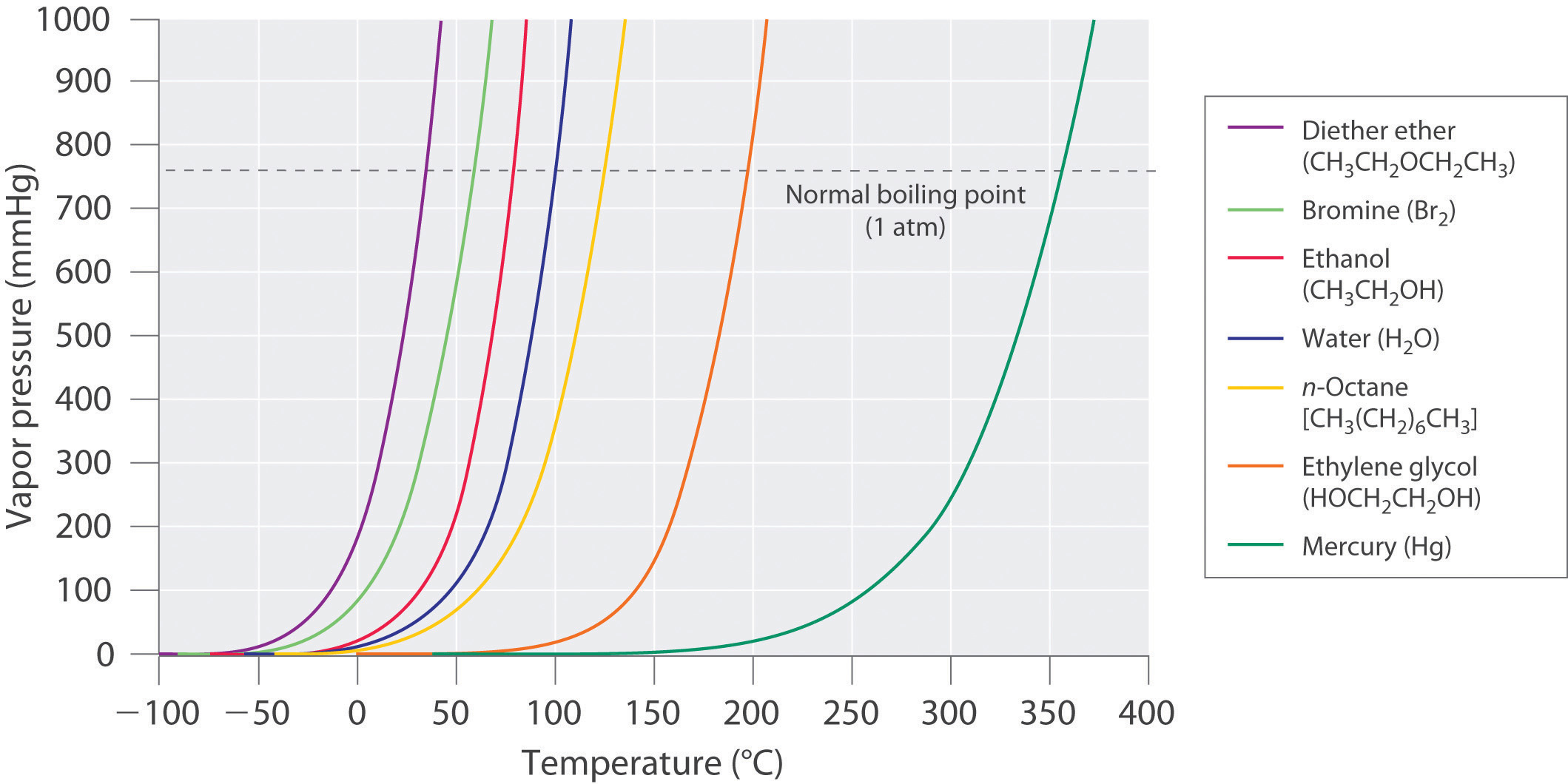
Acetone and carbon disulphide form binary liquid solution showing positive deviation from Raoult's law. The normal boiling point (Tb) of pure acetone is less than that of pure CS2 . Pick out

boiling point of diethyl ether,acetone and water is 35 degree celsius 56 degree Celsius and 100 degree - Brainly.in
Is a 50/50 mixture of acetone and water an azeotrope? Also, why does the first drop of destillate form at 40C? - Quora
How to calculate the boiling point of a mixture made up of 2 liquids ( acetone+water in the same ratios) - Quora

The boiling point of pure acetone is 56.38^(@)C`. When 0.707 g of a compound is dissolved in 10 g of - YouTube

BOILING POINT OF A PURE SUBSTANCE Directions: Graph each table of temperature readings below. Plot the time - Brainly.ph