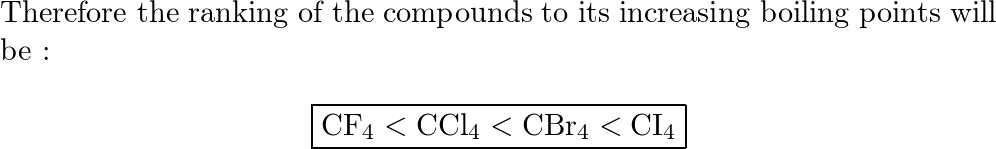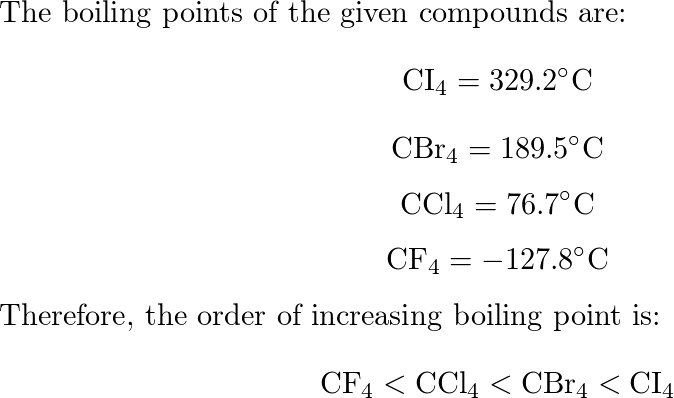SOLVED: Formula CBr4 CH2Br2 Boiling Point (C) 94.5 98.0 Vapor Pressure at 20c (kPa) 5.33 4.65 romide ane ibromomethane has a greater solubility in water than carbon tetrabromide has ccount for this

When the substances CCl4, CH4 , and CF4 are arranged in the order of increasing boiling point, what is the correct order?
What is the order of CH4, CF4, and CCl4 when arranged in order of increasing boiling points? - Quora

SOLVED: In each of the following groups of substances, pick the one that has the given property: highest boiling point: CCl4; CF4, CBr4 b. lowest freezing point: LiF, F2, HCI smallest vapor
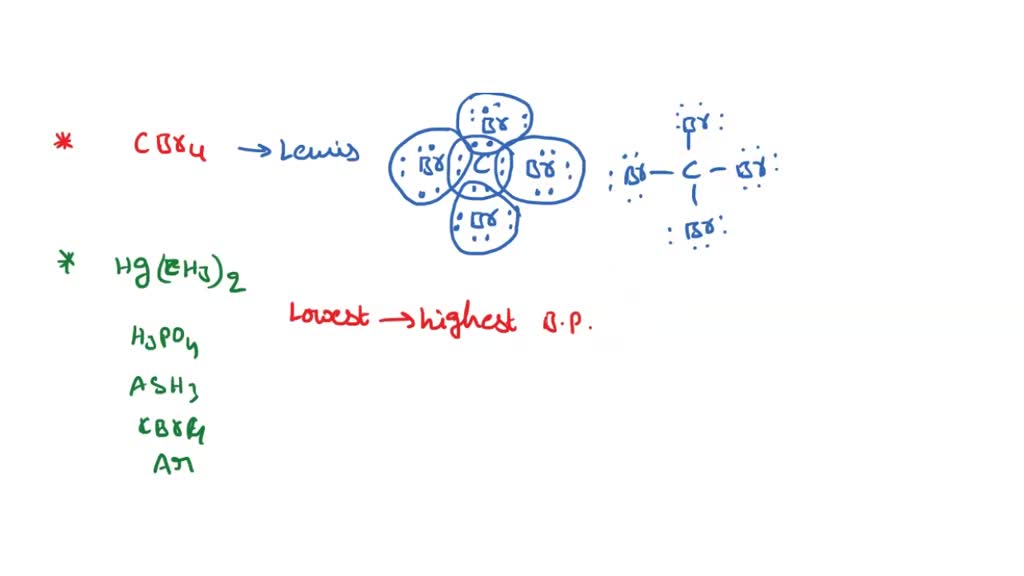
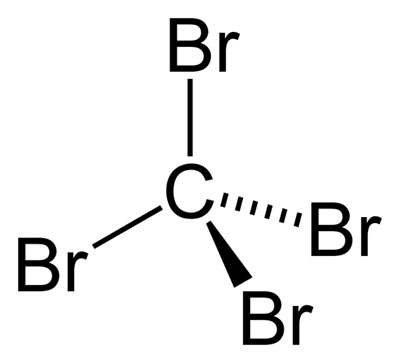
SOLVED: Draw the Lewis structure for carbon tetrabromide, CBr4. Include lone pairs. Arrange the compounds from lowest boiling point to highest boiling point. • Hg(CH3)2 • H3PO4 • AsH3 •CBr4 •Ar

SOLVED: Place the substances in order of decreasing boiling point: BeClz CBr4 BeBrz Select one: BeClz CBra BeBr2 b. BeClz > BeBr2 CBr4 CBra BeBrz BeClz d. BeClz BeBrz CBra BeBr2 BeClz