Enzymes - Tobacco Etch Virus (TEV) and Human RhinoVirus (HRV3C) Cysteine Proteases in Vectors | ATUM - ATUM
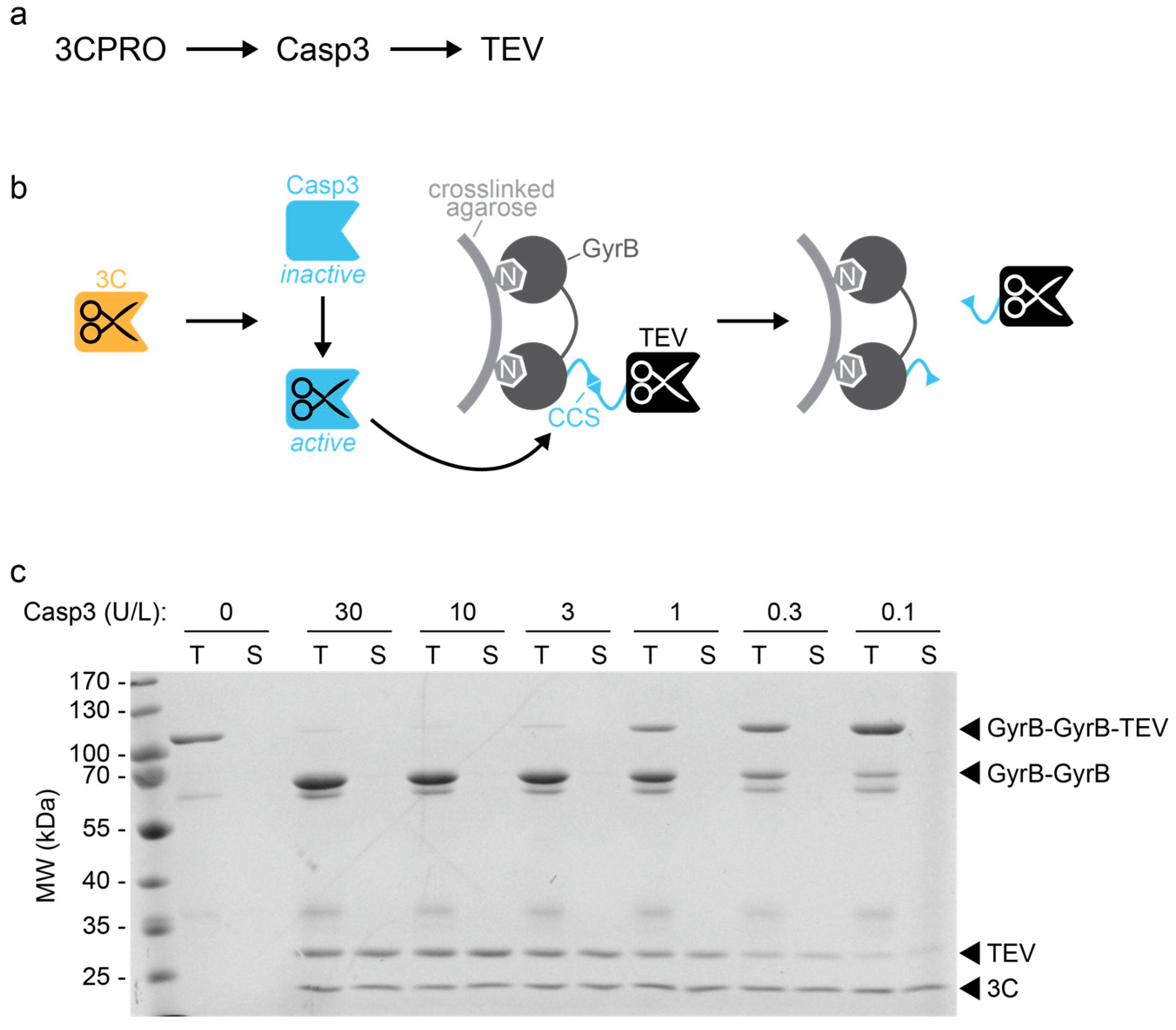
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE

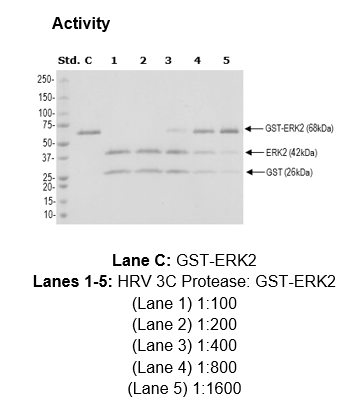
SDS-PAGE showing the HRV 3C protease activity in the presence various... | Download Scientific Diagram

Figure 1 | Expression of the Lactate Dehydrogenase Gene from Eptatretus okinoseanus in Escherichia coli

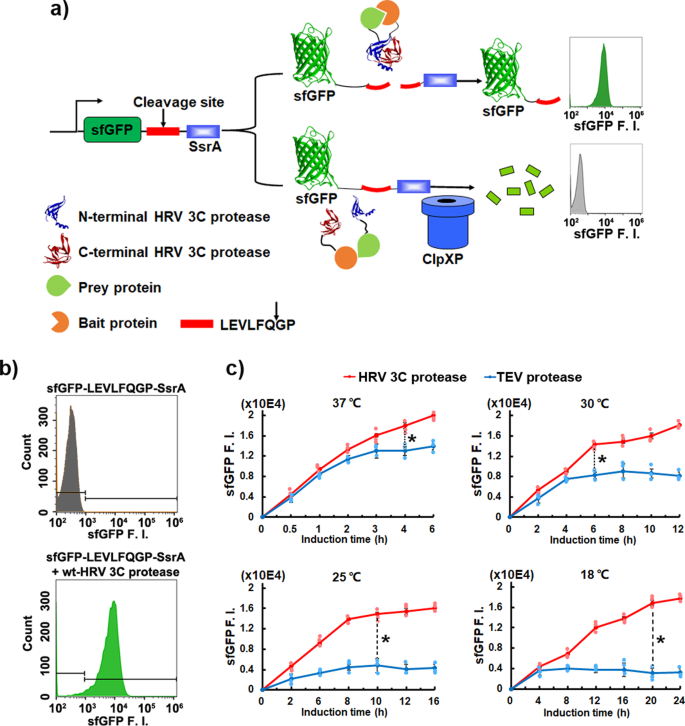
Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE