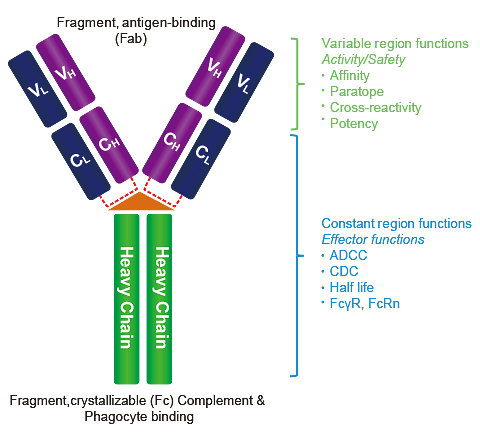
Schematic representation of homodimeric human IgG1-Fc (PDB-ID 1OQO),... | Download Scientific Diagram

Biochemistry, structure, and cellular internalization of a four nanobody‐bearing Fc dimer - Chabrol - 2021 - Protein Science - Wiley Online Library

Structure of the Human IgE-Fc Cε3-Cε4 Reveals Conformational Flexibility in the Antibody Effector Domains - ScienceDirect
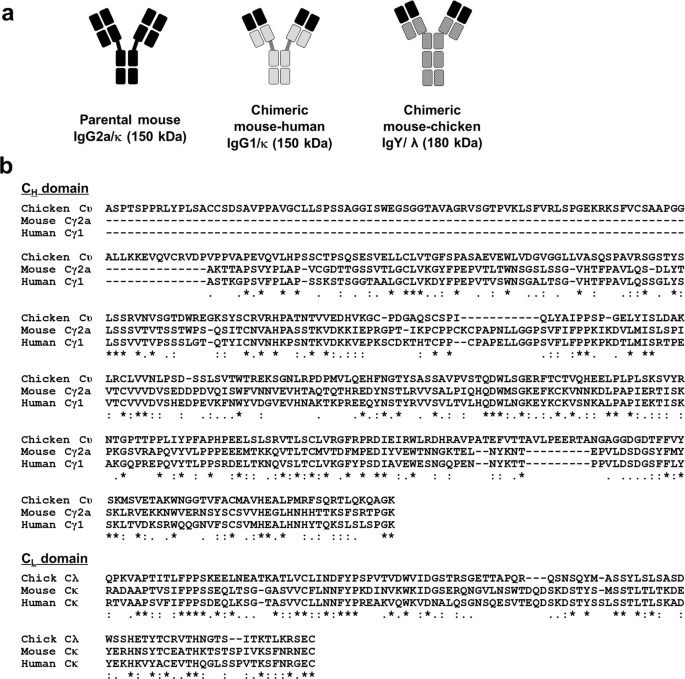
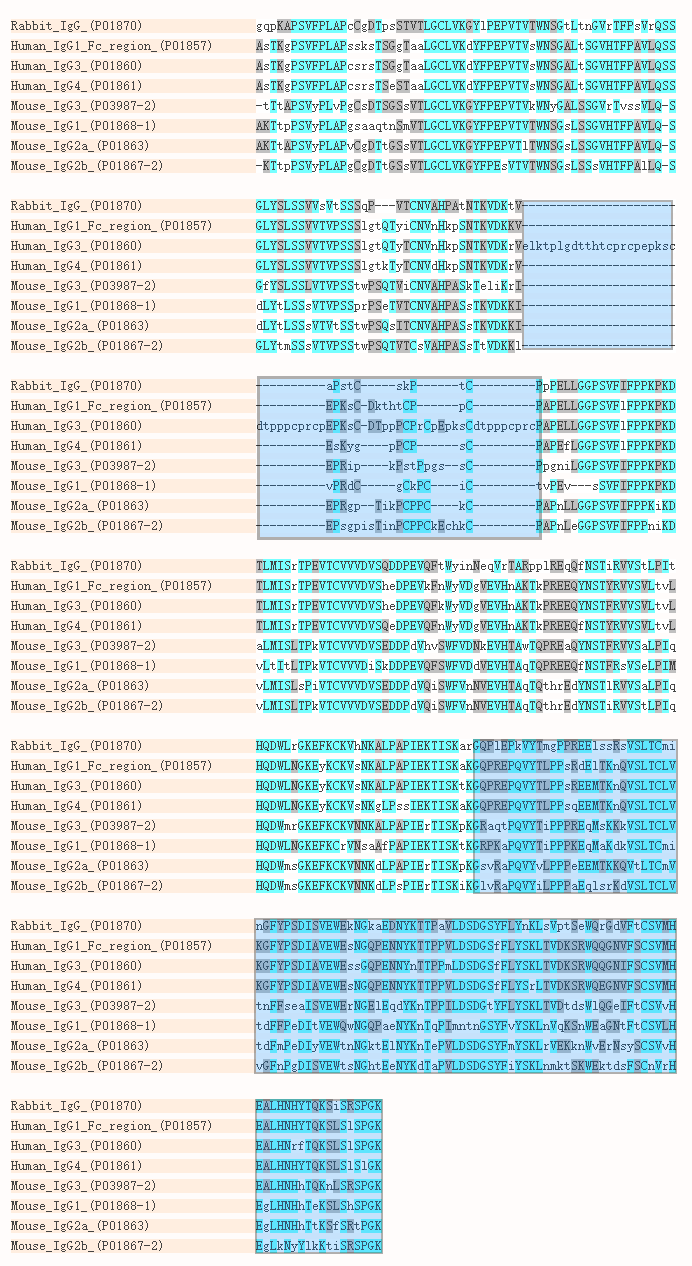
Antigen-binding affinity and thermostability of chimeric mouse-chicken IgY and mouse-human IgG antibodies with identical variable domains | Scientific Reports
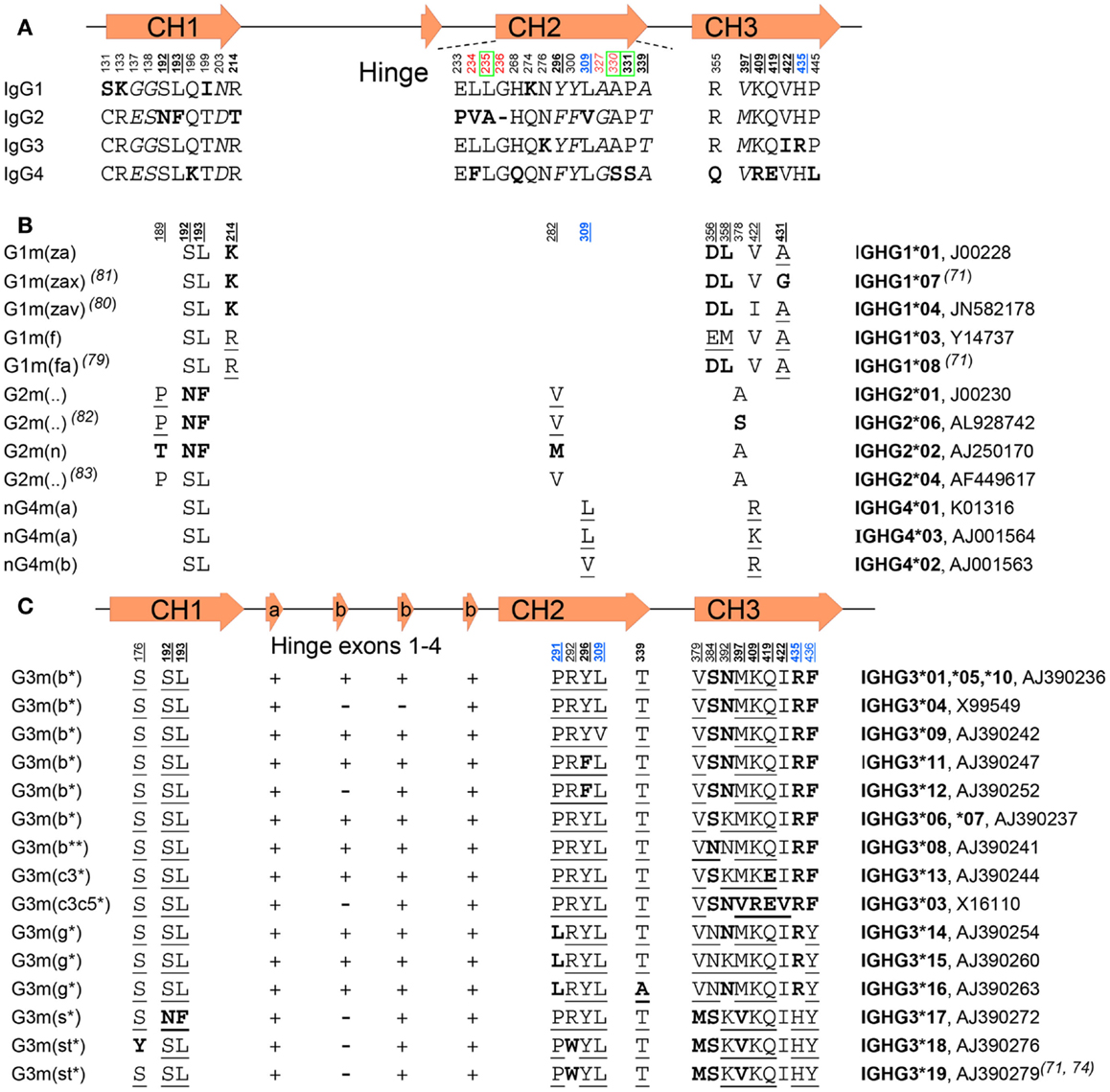
![PDF] The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar PDF] The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/9e5f002374ecf7abd15e4aeacda63aa75162f32a/6-Figure1-1.png)
PDF] The Origins, Specificity, and Potential Biological Relevance of Human Anti-IgG Hinge Autoantibodies | Semantic Scholar
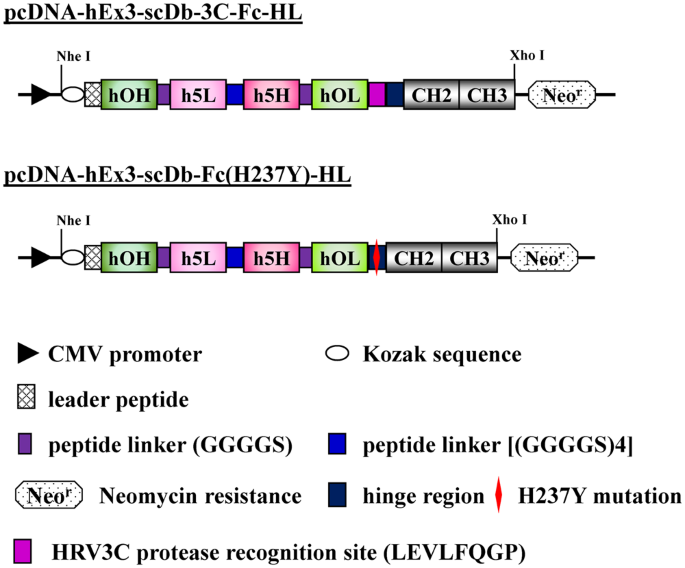
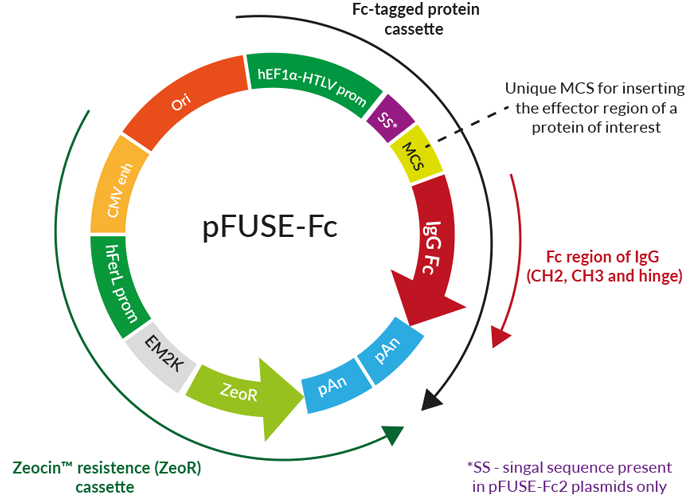
Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports

Engineering the hinge region of human IgG1 Fc-fused bispecific antibodies to improve fragmentation resistance | Scientific Reports

Staphylococcal protein A inhibits complement activation by interfering with IgG hexamer formation | PNAS

Figure 1 from Functional, Biophysical, and Structural Characterization of Human IgG1 and IgG4 Fc Variants with Ablated Immune Functionality | Semantic Scholar

Production of IgG1-based bispecific antibody without extra cysteine residue via intein-mediated protein trans-splicing | Scientific Reports