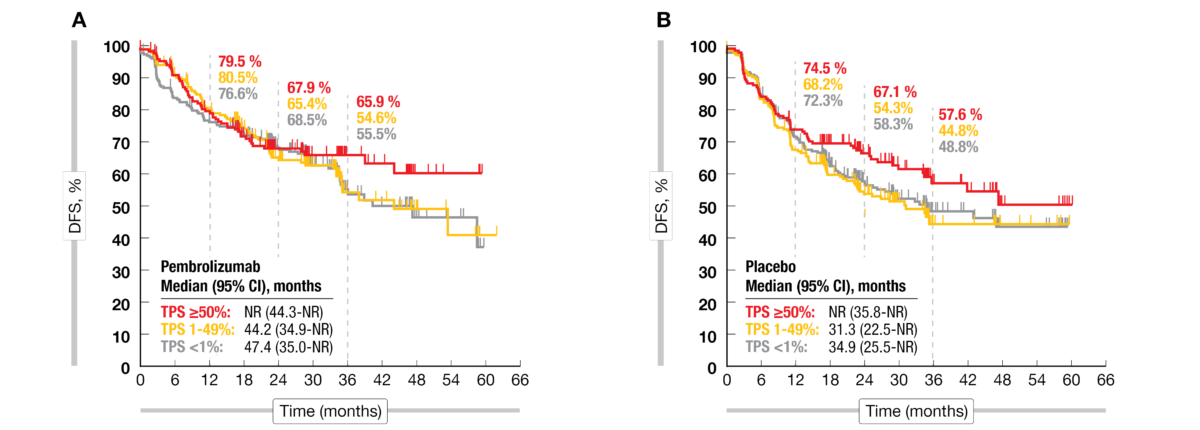
Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial - The Lancet Oncology

Schema of the ACCIO trial including three arms. Pembrolizumab dosing is... | Download Scientific Diagram

Oncology Brothers on Twitter: "1. #KN091 #PEARLS by @peters_solange : Pembrolizumab in adjuvant settings and after chemotherapy for stage IB (T2a ≥4 cm), II, or IIIA NSCLC. Approved for PDL1 expressions. -
Frühes nichtkleinzelliges Bronchialkarzinom: Pembrolizumab adjuvant nach Resektion verlängert das krankheitsfreie Überleben

Stephen V Liu, MD on Twitter: "#ASCO22 Discussion of PEARLS (KEYNOTE-091, @ETOP_eu 8-15), a phase III study of adjuvant pembrolizumab in resected NSCLC. The study did meet its primary endpoint (DFS HR
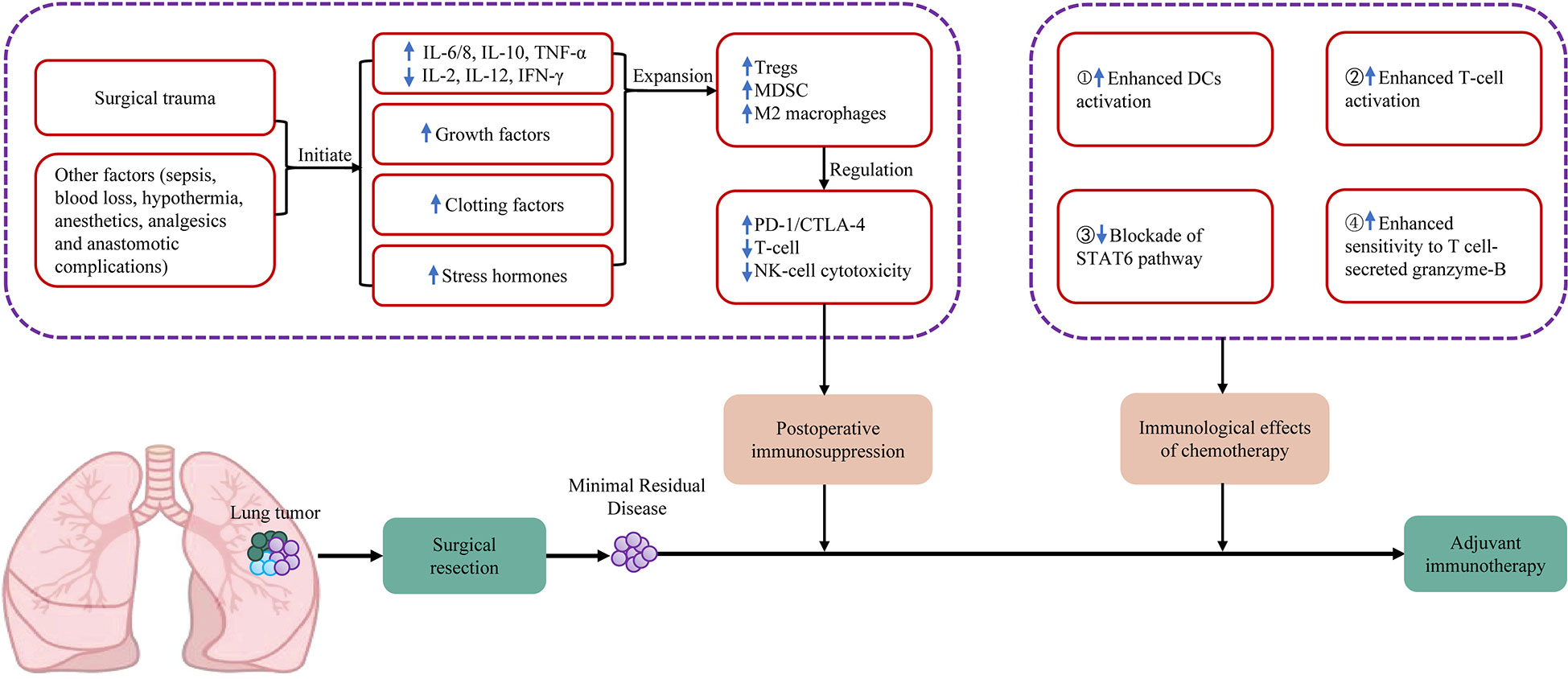
Frontiers | Adjuvant immunotherapy in early-stage resectable non–small cell lung cancer: A new milestone
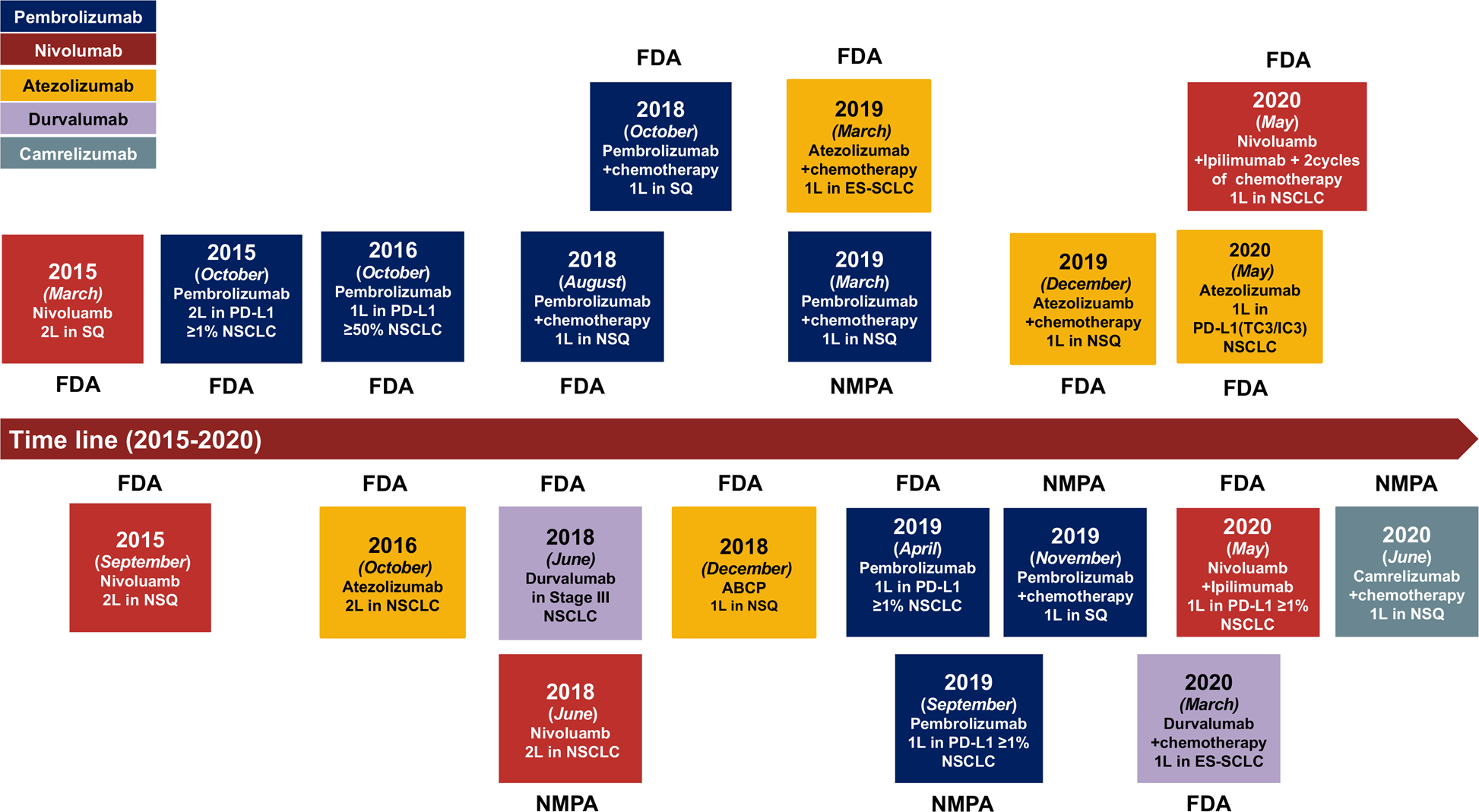
The cutting-edge progress of immune-checkpoint blockade in lung cancer | Cellular & Molecular Immunology

Aakash Desai, MD, MPH on Twitter: "@LuisPaz_Ares discussed data from #PEARLS study #Pembrolizumab for early stage #NSCLC #LCSM with dual primary endpoint of DFS in all comers/PDL1>50%, showing significant DFS improvement HR:0.76
Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial | AIOM

Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial | AIOM

Immune Checkpoint Inhibitors in Thoracic Malignancies: Review of the Existing Evidence by an IASLC Expert Panel and Recommendations - ScienceDirect