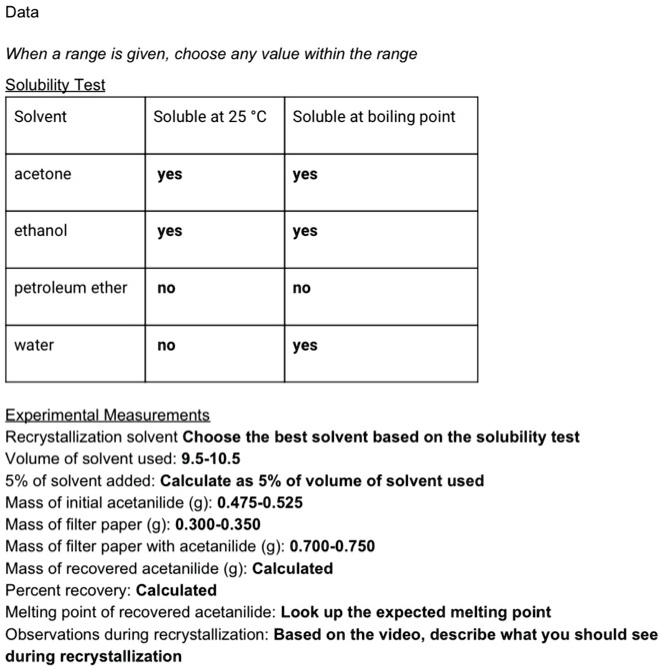
![8032-32-4・Petroleum Ether・168-00815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation 8032-32-4・Petroleum Ether・168-00815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation](https://labchem-wako.fujifilm.com/sc/05/168-00815.png)
8032-32-4・Petroleum Ether・168-00815[Detail Information]|Laboratory Chemicals-FUJIFILM Wako Chemicals U.S.A. Corporation

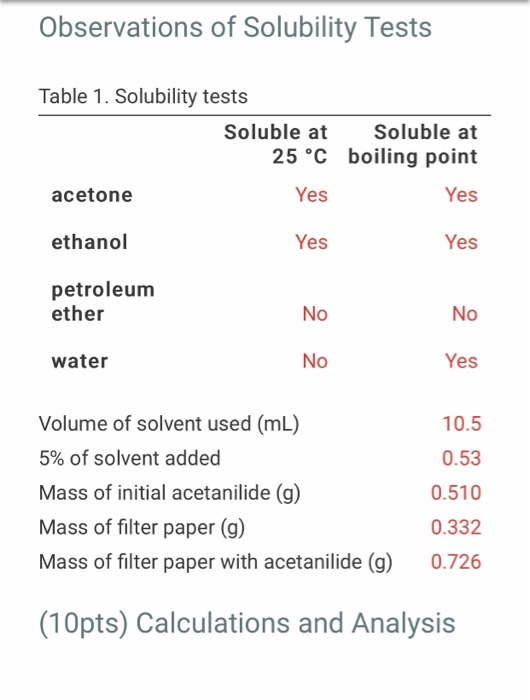
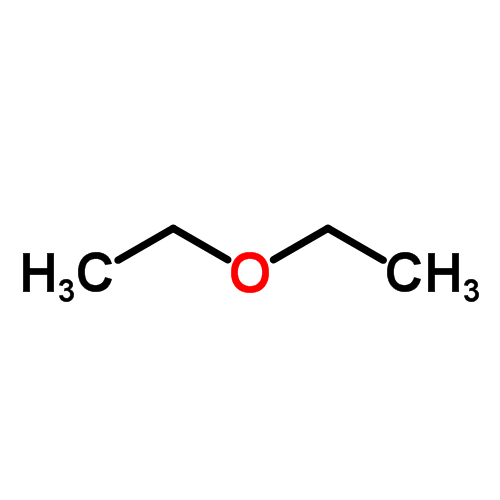
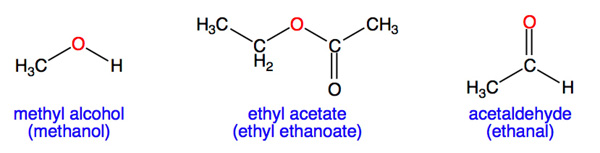
The boiling point of butan-2-one (80 C) is significantly higher than the boiling point of diethyl ether (35 C), even though both compounds exhibit dipole-dipole interactions and have comparable molecular weights. Offer
Petroleum ether 40-60 °C, 5 l, aluminium | Acid Value Determination | Analysis of Food | Inorganic & Analytical Reagents | Chemicals | Carl Roth - International
Why is ethanol boiling at higher boiling point than diethyl ether which boils at a low boiling point yet they have the same molecular weight? - Quora