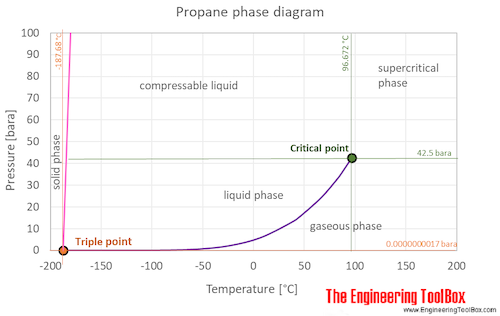
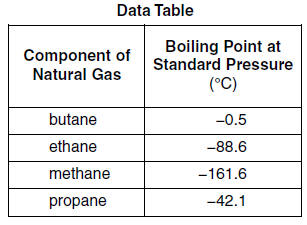
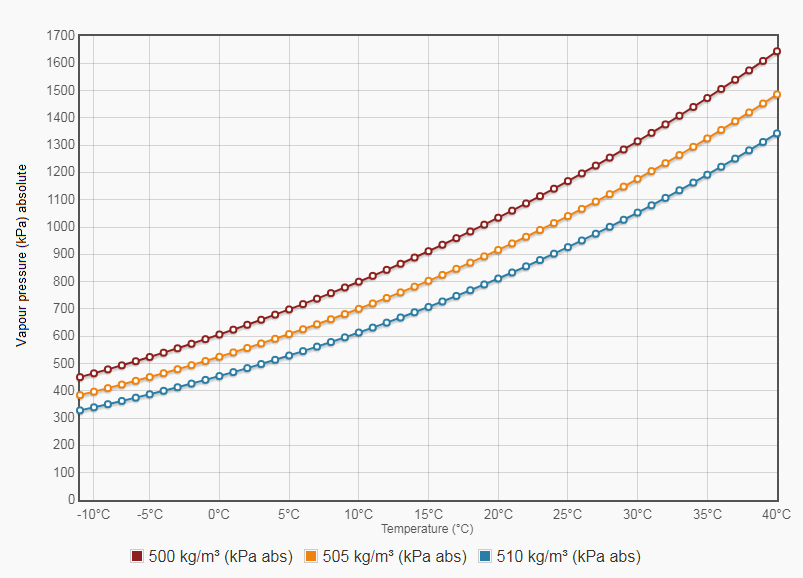
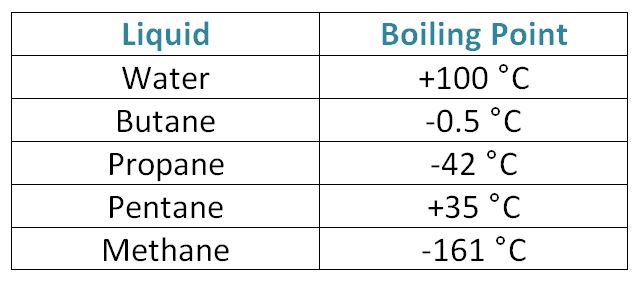
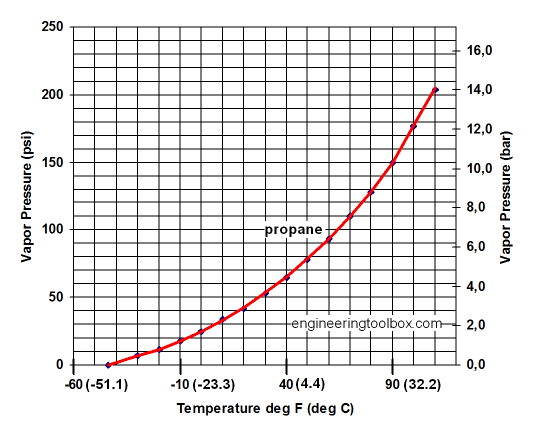
Oil and Gas Training Tip: Everything Will Make More Sense if You Know This About Hydrocarbons - Energy Training Resources

Why is the boiling point of propane-1,3-diol (HOCH_2CH_2CH_2OH) higher than the boiling point of propane-1,2-diol (HOCH_2CH(OH)CH_3) (215 degrees C vs. 187 degrees C)? Why do both diols have a higher boiling point
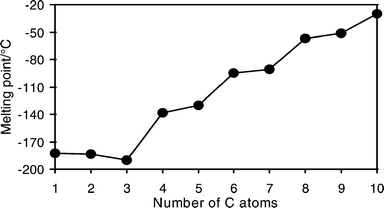
Why is the melting point of propane the lowest among n -alkanes? - New Journal of Chemistry (RSC Publishing) DOI:10.1039/B004283H
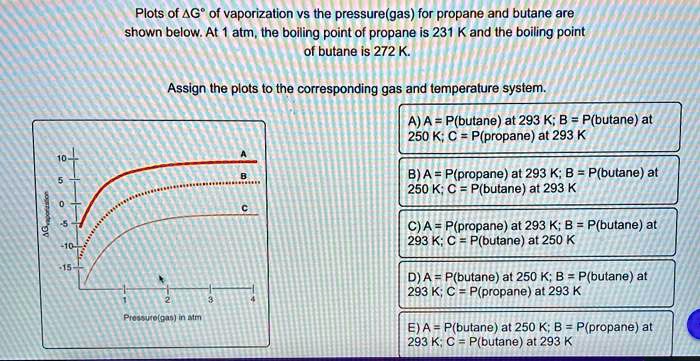
SOLVED: Plots of AG? of vaporization vs the pressure(gas) for propane and butane are shown below: At atm , the boiling point of propane is 231 Kand Ihe boiling point of butane